TESTING PLANT SUBSTANCES AS POTENTIAL MEDICINES
Background: Plants, like any other organism, have to compete with each other for resources, like water, space, and sunlight. Plants have developed different characteristics to give them advantages in competition: big leaves, height, and toxic chemicals that they excrete to kill surrounding plants.
Organisms all can be infected by viruses or threatened by bacterial diseases. Some organisms have an immunity to the "foreign" invaders. Their immune system consist of antimicrobial agents that fight the microbes of the diseases. Finding and isolating the antimicrobial molecules can lead to the discovery of potential medicine.
With different processes, bio-technicians extract and sample plant substances to see if they kill microbes and don't cause toxic effects in humans. After several trials, bio-technicians can determine whether the plant nutrients and chemicals can cause bacterial death.
Objective/Purpose: What local plant materials contain active ingredients will inhibit the growth of bacteria?
Materials: ~balance, weigh boat, lab scoops ~inoculating loop, Ni/Cr wire ~syringe, 10 mL and filter
~LB broth base ~petri dishes, 60x15 mm, sterile ~reaction tubes and rack
~media bottles, 250 mL ~E.Coli JM109 (stock plate) ~methanol, absolute
~sterilizer/autoclave ~plant specimen ~pipet, 1 mL and pump
~water bath, 37*C, shaking ~mortar and pestle ~dry block heater
~sterile LB agar ~pipet, 10 mL and pump ~forceps, fine-tipped
~laminar flow hood and disinfectant ~plastic funnels, short stemmed ~ampicillin
~glasses, safety, plastic ~filter paper disks, 5mm diameter ~glass spreader
~Bunsen burner and gas lighter ~beakers, 100mL ~incubator oven, 37*C
Procedure:
Preparing the plant extracts:
- using a mortar and pestle, grind up two grams of plant tissue with 10mL of deionized water
- let it set for three minutes
- filter the sample through am 11 cm filter paper/funnel
- filter sterilize the extract using a syringe filter
- collect 1 mL of the filter-sterilized extract into a 1.7 microbe
- label the sample
- repeat steps, but substitute water with methanol
- attach pre-filter to syringe and rinse with water
- take your plant extract, syringe/pre-filter, and pipet to the Laminar Hood
- label microfuge tube with "water" or "methanol", depending in which extract you are doing first
- attach the sterile filter to the pre-filter (DON'T TOUCH THE STERILE FILTER, EVEN WITH GLOVES!!!!!)
- load 1.7 mLs of the extract into the syringe, using the pipet
- press the plunger into 1.0 mL filter of the sterilized extract
- place a cap on the microfuge tube
- evaporate methanol from methanol extract by placing the tube (with the cap open) on a 65*C heat block overnight.
- reconstitute the methanol extract with 1.0 mL of sterile deionized water
- using sterile forceps, place three pieces of filter paper in each extract tube
- store the tubes at 4*C until they are ready to use
- draw a plus sign on the bottom of each plate, and number the quadrants one through four
- liquefy the sterile LB agar in the microwave
- using sterile techniques, pour approximately 20 mL of agar into the petri plate
- transfer 1mL of E.Coli broth (provided by the teacher) to the middle of each petri dish holding the solutions
- sterilize a spreading loop using alcohol and flame
- use the spreading loop to spread the E.Coli around the petri plates. Cover immediately after, and let soak for 15 minutes
- place sterile disks to the appropriate tubes of filtered extract (two disks in sterile water, two disks in ampicillin)
- place the sterile disks in the appropriate solution in the agar plate
- incubate the solutions at 37*C for 24 to 48 hours
Results and Observations:
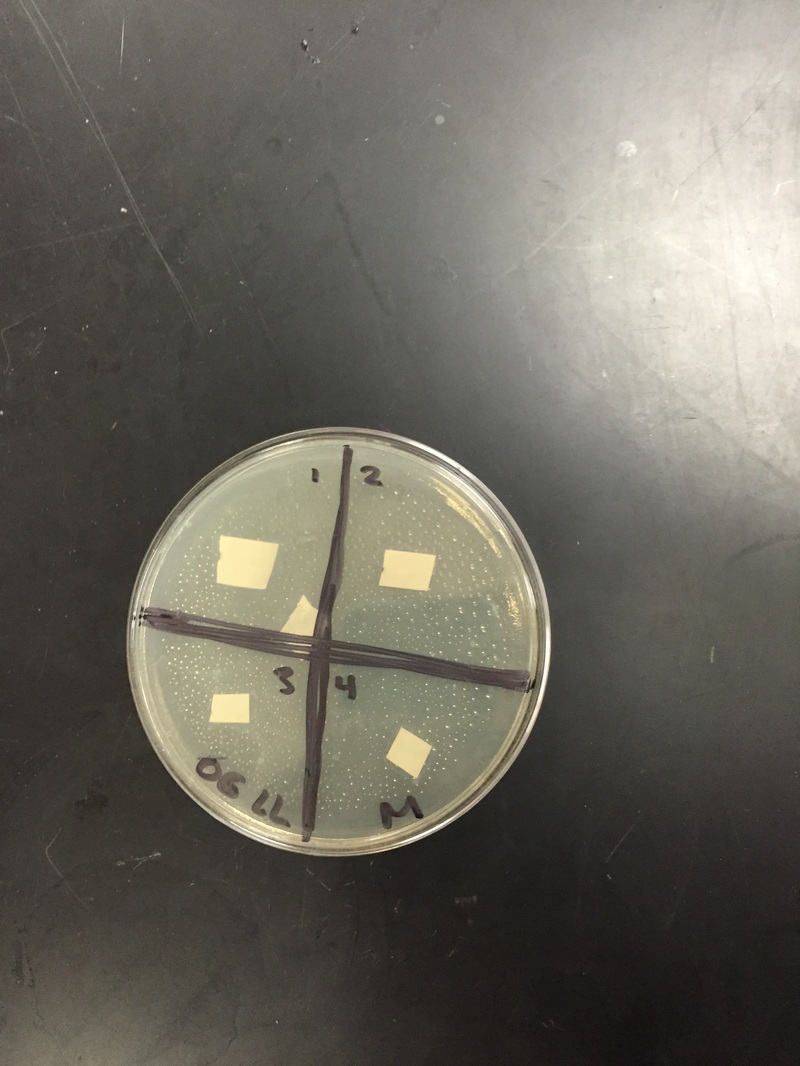
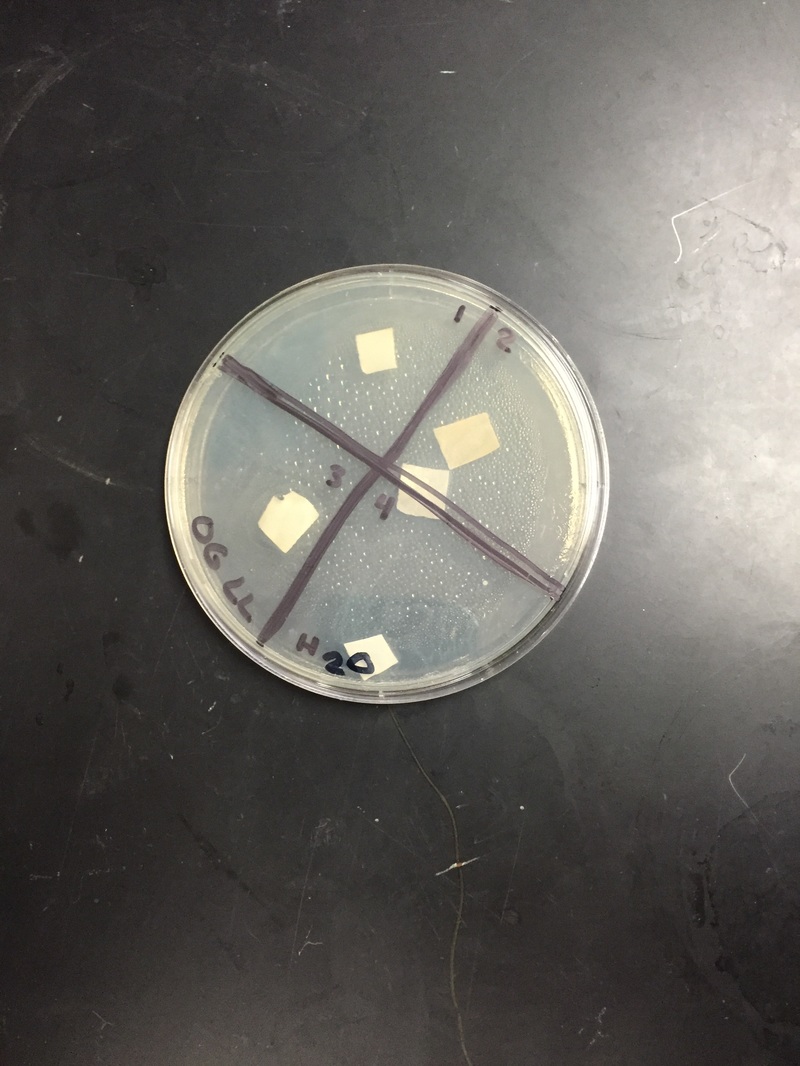
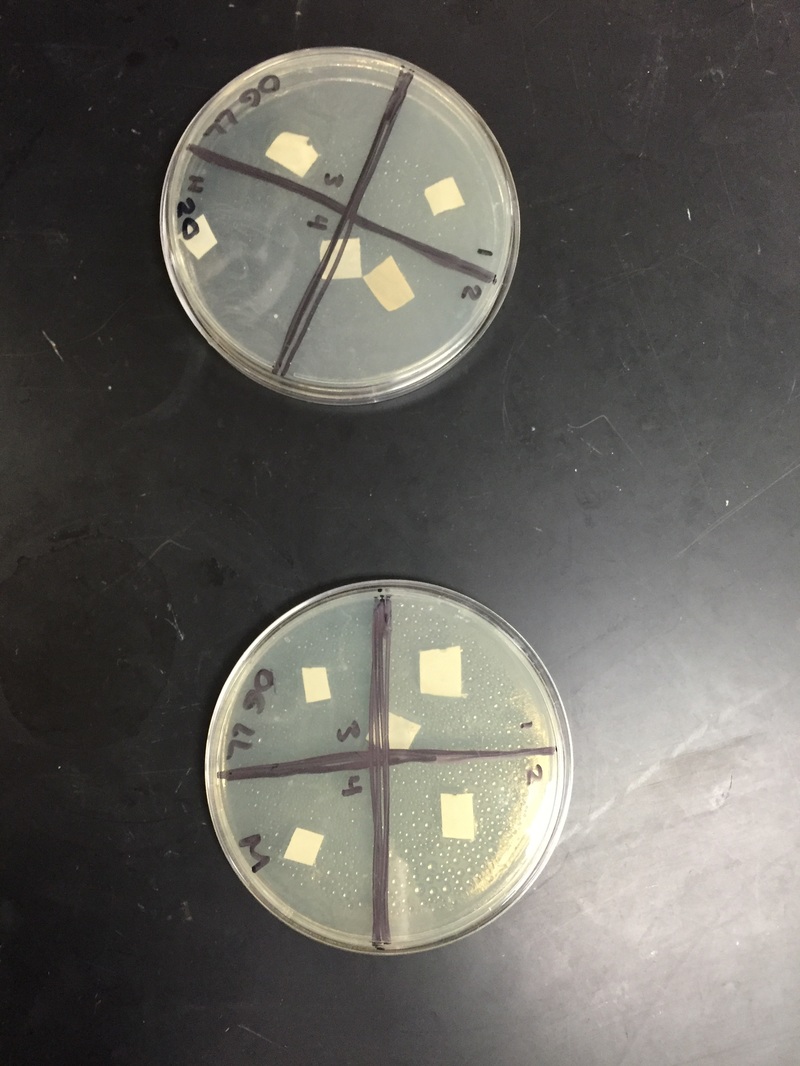
day 1: We looked at our petri dishes to see if the bacteria grew and showed the results; the results being whether there was a clearance around our paper disks. We didn't see too much of a difference, and you could barely tell whether there was a clearance or not. We decided to wait another day.
day 2: We saw a small difference from day one. It was hard to decipher the difference between the cloudy bacteria and the clear agar. There was a small clearance around the disks, but like said above, it was hard to tell. We are waiting one more day before we write our results.
Analysis and Conclusion
Our results ended up being inconclusive; we couldn't tell if the bacteria grew at all. It was very hard to see the clearance.
Our results ended up being inconclusive; we couldn't tell if the bacteria grew at all. It was very hard to see the clearance.